A battery stores and supplies electrical energy to various systems in a vehicle, especially before the engine starts. It is a critical component that enables the vehicle to begin operation properly.
Functions of a Battery
1. Engine Starting
The battery supplies electrical current to the starter motor, which turns the engine to initiate operation. Electrical energy is converted into mechanical energy until the engine starts and runs independently.
2. Supplying Power to Electrical Devices
The battery provides electrical power to various components in the vehicle, such as lighting systems, signaling systems, and electronic devices—particularly when the engine is not yet running or when the power generation system is insufficient.
3. Maintaining Voltage Stability
The battery helps absorb and regulate voltage fluctuations within the electrical system, ensuring a stable power supply. This protects electrical components and electronic systems, allowing them to operate efficiently.
Types of Batteries
Batteries can be classified based on the type of electrolyte and their usage characteristics. Each type has distinct properties and limitations, as follows:
Classification by Electrolyte Type
Dry Cell: Uses a semi-solid or paste-like electrolyte, with no free liquid that could leak. It can operate in various orientations and is suitable for general portable devices.
Wet Cell (Storage Battery): Uses a liquid electrolyte and can be recharged and reused. A common example is the lead-acid battery used in cars and motorcycles.
Classification by Usage Characteristics
Primary Battery: A non-rechargeable battery that must be replaced once depleted. Examples include alkaline batteries and lithium batteries used in small electronic devices.
Secondary Battery: A rechargeable battery that can be used multiple times. Examples include lithium-ion batteries in mobile phones and computers, as well as batteries used in vehicles.
Secondary batteries can be found in both dry cell and wet cell forms. Common types include lead-acid batteries, lithium/lithium-ion batteries, and nickel-based batteries. Each type operates based on different chemical principles and is suited to different applications.
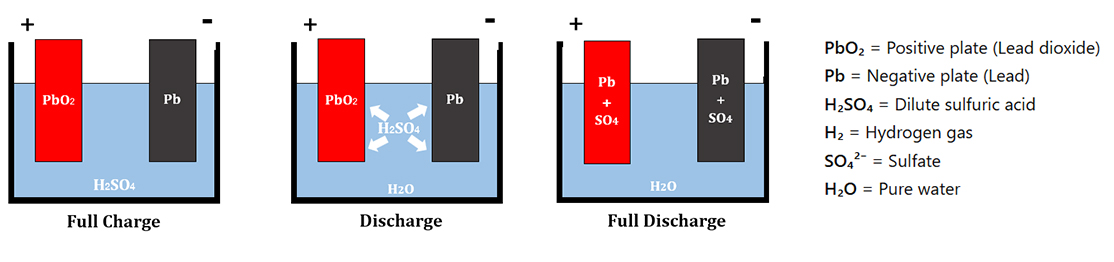
Working Principle of a Lead-Acid Battery
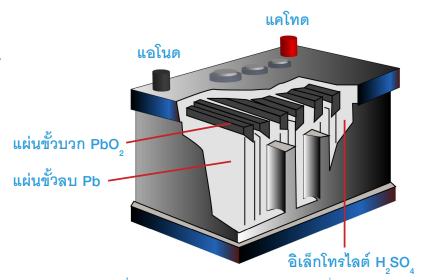
A Lead-Acid Battery typically consists of 6 cells, each producing about 2 volts, resulting in a total of approximately 12 volts. The electrodes are made of lead.During charging, the lead plate connected to the positive terminal reacts with the electrolyte, which is sulfuric acid (H₂SO₄). This reaction produces Pb²⁺ ions, which then combine with oxygen to form lead (IV) oxide (PbO₂), as represented by the chemical equation.
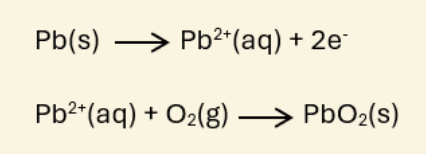
After that, the lead plate coated with PbO₂ acts as the anode, while the other lead plate acts as the cathode. The electrochemical reactions that occur during discharging (supplying electrical current) and charging are as follows:

Chemical Reaction Principles of a Lead-Acid Battery